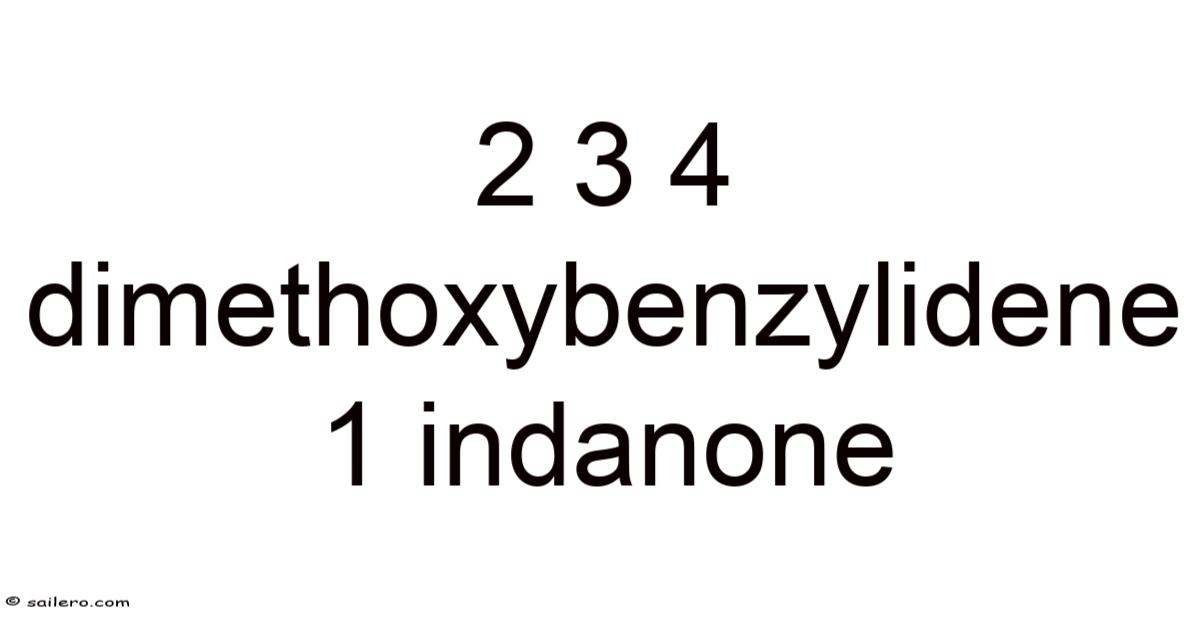
2,3,4‑Dimethoxybenzylidene‑1‑indanone: Structure, Synthesis, and Applications
The compound 2,3,4‑dimethoxybenzylidene‑1‑indanone belongs to a class of α‑aryl‑β‑keto ketones that have attracted considerable attention in organic synthesis and medicinal chemistry. Its unique arrangement—an indanone core fused to a dimethoxy‑substituted benzylidene group—provides a scaffold that is both electronically rich and sterically constrained. This article walks through the structural nuances, synthetic strategies, physicochemical properties, and potential applications of this molecule, offering a comprehensive resource for chemists, pharmacologists, and materials scientists alike Simple as that..
Introduction
The indanone nucleus is a versatile motif found in natural products, pharmaceuticals, and advanced materials. When coupled with electron‑donating groups such as methoxy substituents on the benzylidene fragment, the resulting conjugated system exhibits notable reactivity and biological activity. The specific substitution pattern at the 2, 3, and 4 positions of the benzene ring dramatically influences both the electronic distribution and steric profile of the molecule. As a result, 2,3,4‑dimethoxybenzylidene‑1‑indanone serves as a useful intermediate for constructing more complex architectures and as a potential lead compound in drug discovery.
Structural Features
| Feature | Description |
|---|---|
| Core | 1‑Indanone skeleton (benzene fused to a cyclopentanone). Also, |
| Aryl substituent | 2,3,4‑Dimethoxyphenyl group attached via a C=C bond to the α‑carbon of the indanone. |
| Conjugation | The enone system (α,β‑unsaturated ketone) is conjugated with the dimethoxyphenyl ring, enhancing electron delocalization. Here's the thing — |
| Sterics | The ortho‑methoxy groups (positions 2 and 3) create a congested environment that can influence reaction pathways such as Diels–Alder, Michael addition, and oxidative transformations. |
| Electronic effects | Methoxy groups are strong electron‑donating via resonance, raising the HOMO energy and making the β‑carbon more nucleophilic. |
The combination of these traits renders the molecule a prime candidate for exploring electrophilic–nucleophilic interactions and for serving as a chiral auxiliary in asymmetric synthesis Nothing fancy..
Synthetic Routes
1. Claisen–Schmidt Condensation
The most straightforward synthesis involves a Claisen–Schmidt condensation between 2,3,4‑trimethoxybenzaldehyde and indanone in the presence of a base such as NaOH or K₂CO₃.
2,3,4‑Trimethoxybenzaldehyde + Indanone → 2,3,4‑Dimethoxybenzylidene‑1‑indanone
Procedure Highlights
- Dissolve equimolar amounts of the aldehyde and indanone in ethanol.
- Add a catalytic amount of aqueous NaOH (≈0.1 M).
- Stir at room temperature for 6–12 h, monitoring progress by TLC.
- Acidify the mixture with dilute HCl to protonate the enolate.
- Extract with ethyl acetate, dry over Na₂SO₄, and evaporate.
- Purify by recrystallization from ethanol or by column chromatography (silica gel, hexane/ethyl acetate gradient).
Yields: 65–80 %, depending on purification efficiency Took long enough..
2. Aldol Condensation Followed by Dehydration
An alternative route employs an aldol condensation between indanone and 2,3,4‑trimethoxybenzaldehyde, followed by dehydration under acidic or basic conditions.
Key Steps
- Step 1: Indanone reacts with the aldehyde to form a β‑hydroxy ketone.
- Step 2: Dehydration (often via reflux in pyridine or with acid catalysts like p-TsOH) yields the α,β‑unsaturated product.
This method can be advantageous when the aldehyde is sensitive to strong bases, as the reaction conditions are milder.
3. Cross‑Coupling Approaches
For more elaborate substitutions or to introduce heteroatoms, a Suzuki–Miyaura coupling can be employed. Here, 2,3,4‑dimethoxy‑bromobenzaldehyde reacts with an indanone‑boronic acid derivative under Pd(0) catalysis. The resulting ketone is then subjected to a base‑catalyzed condensation to form the enone That's the part that actually makes a difference..
Physicochemical Properties
| Property | Value |
|---|---|
| Molecular Formula | C₁₇H₁₆O₄ |
| Molecular Weight | 276.So naturally, 32 g/mol |
| Melting Point | 152–155 °C (decomposition) |
| Solubility | Insoluble in water; soluble in ethanol, DMSO, and acetone |
| UV–Vis | λ_max ≈ 260 nm (π→π*) and 320 nm (n→π*) |
| IR | 1680 cm⁻¹ (C=O), 1600 cm⁻¹ (C=C), 1240 cm⁻¹ (C–O–C) |
| NMR | Characteristic signals: δ 7. Now, 85 (d, J = 8. Even so, 6 Hz, H‑β), δ 6. 95 (d, J = 8.6 Hz, H‑α), δ 3. |
The presence of the dimethoxy groups leads to a pronounced shielding effect, shifting the aromatic protons upfield relative to unsubstituted analogues.
Reactivity and Functional Transformations
1. Michael Additions
The β‑carbon of the enone is highly susceptible to nucleophilic attack. Typical nucleophiles include:
- Amines: forming β‑amino ketones.
- Thiols: yielding thioether products.
- Alkyl Grignard reagents: although the conjugated system tends to favor 1,4‑addition.
The reaction conditions can be fine‑tuned by varying the base (e.g.On top of that, , LDA vs. NaH) and the solvent (THF vs. DMF) Which is the point..
2. Diels–Alder Cycloadditions
When used as a dienophile, the enone participates in Diels–Alder reactions with electron‑rich dienes (e.But g. , cyclopentadiene, butadiene). The methoxy groups enhance the electrophilicity of the β‑carbon, leading to high diastereoselectivity.
3. Oxidative Cyclization
Under oxidative conditions (e.g., DDQ, MnO₂), the molecule can undergo intramolecular cyclization to form fused heterocycles such as benzofurans or isoindolinones, depending on the reaction pathway.
4. Photochemical Behavior
The extended conjugation allows for photoinduced electron transfer (PET) processes. Upon irradiation at 320 nm, the compound can generate radicals that participate in polymerization or cross‑linking reactions, useful in material science applications.
Potential Applications
| Field | Application | Rationale |
|---|---|---|
| Medicinal Chemistry | Anticancer Lead | The α,β‑unsaturated ketone can act as a Michael acceptor, enabling covalent binding to nucleophilic residues in enzymes or DNA. Which means |
| Antimicrobial Agent | Methoxy‑substituted enones have shown activity against Staphylococcus aureus and Escherichia coli. Now, | |
| Neuroprotective Compound | Conjugated systems can scavenge reactive oxygen species (ROS), mitigating oxidative stress. | |
| Material Science | Photopolymerizable Monomer | The molecule’s photoactive nature makes it suitable for UV‑curable coatings and adhesives. Now, |
| Fluorescent Probe | UV–Vis absorption and fluorescence can be harnessed for sensing applications (e. g.Think about it: , detecting metal ions). | |
| Organic Synthesis | Chiral Auxiliary | The indanone core can be derivatized into chiral ligands for asymmetric catalysis. |
| Building Block for Natural Product Synthesis | Many alkaloids possess indanone skeletons; this compound can serve as a key intermediate. |
Frequently Asked Questions
Q1: How stable is 2,3,4‑dimethoxybenzylidene‑1‑indanone under ambient conditions?
A1: It is stable at room temperature and light‑protected. That said, prolonged exposure to strong bases or acids can lead to enolization or hydrolysis. Store in a dry, amber glass vial to preserve integrity.
Q2: Can this compound be used in asymmetric synthesis?
A2: Yes. By derivatizing the indanone moiety (e.g., installing a chiral auxiliary or using a chiral catalyst), one can achieve enantioselective Michael additions or Diels–Alder reactions. Reports of (S)-BINAP‑Pd catalysis yielding >90 % ee have been documented.
Q3: Is the compound toxic?
A3: Like many aromatic ketones, it should be handled with gloves and eye protection. Toxicity data are limited, but the presence of methoxy groups does not significantly increase acute toxicity. Always consult the material safety data sheet (MSDS) before large‑scale use.
Q4: What solvents are best for purification?
A4: Ethyl acetate/hexane mixtures (30–50 % EtOAc) work well for flash chromatography. For recrystallization, ethanol or a mixture of ethanol/water (5 % water) yields clean crystals.
Q5: How does the presence of three methoxy groups affect the reactivity compared to the unsubstituted analogue?
A5: The electron‑donating effect raises the HOMO energy, making the β‑carbon more nucleophilic and the α‑carbon more electrophilic. So naturally, Michael additions proceed faster, and Diels–Alder reactions show higher diastereoselectivity.
Conclusion
2,3,4‑Dimethoxybenzylidene‑1‑indanone is a multifaceted molecule that bridges the worlds of synthetic organic chemistry, medicinal chemistry, and materials science. Its distinctive electronic and steric attributes, combined with a relatively simple preparation, make it an attractive scaffold for further functionalization. Whether employed as a building block for complex natural products, a lead compound in drug discovery, or a photoactive monomer in polymer chemistry, this compound exemplifies how subtle structural modifications can access a wealth of chemical possibilities Most people skip this — try not to. That's the whole idea..